 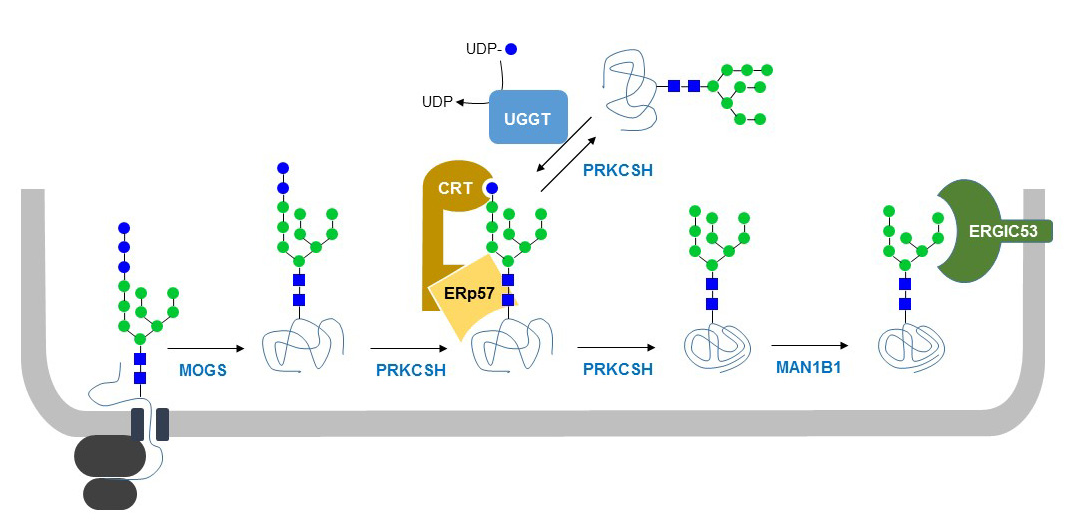
Lectin-glycoprotein association not only thwarts Golgi exit of folding intermediates and irreparably misfolded glycoproteins but also enhances folding efficiency by preventing aggregation and promoting proper disulfide bonding. Alternatively, permanently misfolded glycoproteins may be then transported to the cytosol for proteasomal degradation. Glucose-free glycoproteins then continue their transit through the secretory pathway. Cycles of CNX/CRT-glycoprotein binding and liberation, catalyzed by the opposing activities of GT and GII, are terminated once glycoproteins attain their native structures. The proteinlinked glycan is then reglucosylated by the soluble ER enzyme GTonly if the protein moiety displays non-native three-dimensional structures, as this enzyme behaves as a conformational sensor. Calnexin and calreticulin are related proteins that comprise an ER chaperone system that ensures the proper folding and quality control of newly synthesized glycoproteins. This is followed by removal of the innermost glucose by GII, thus liberating the glycoprotein from the lectin anchor. This structure is then recognized by two ER resident lectins (CNX and CRT) that specifically bind monoglucosylated polymannose glycans. 1) (1–3).2 Glycan processing starts immediately after its transfer from a dolichol-P-P derivative to Asn residues in nascent polypeptide chains entering the lumen of the ER.3 Removal of the outermost and following glucoses by the successive action of GI and GII exposes the Glc1Man9GlcNAc2 epitope (Fig. Roles of calreticulin and calnexin during mucin synthesis in LS180 and HT29/A1 human colonic adenocarcinoma cells 2. The N-glycan-dependent quality control mechanism of glycoprotein folding was proposed initially by Helenius and coworkers several years ago with a few minor modifications, it is still valid today (Fig. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
